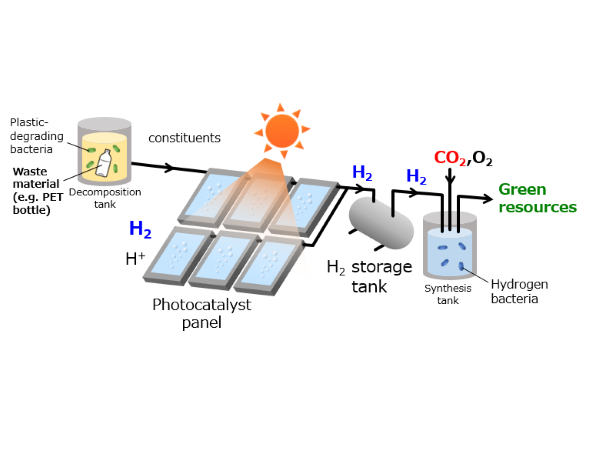
Towards widespread adoption of solar hydrogen production
Using a hematite (*1) photocatalyst (*2), a joint research team has succeeded in producing both hydrogen gas and hydrogen peroxide (*3) at the same time from sunlight and water. The team included the following members from Kobe University: Associate Professor TACHIKAWA Takashi (of the Molecular Photoscience Research Center) Professor TENNO Seiichiro (Graduate School of System Informatics/ Graduate School of Science, Technology, and Innovation), Associate Professor TSUCHIMOCHI Takashi (Graduate School of System Informatics) et al.
In the quest to make a carbon neutral society a reality, CO2-free hydrogen production using sunlight energy has gained attention. If chemical products with applications in the health and food industries could be produced at the same time as hydrogen through photocatalyst-mediated solar water-splitting, it would be possible to develop a solar water-splitting utilization system with even greater added value.
Hematite mesocrystals (*4) can absorb a wide range of visible light. In this study, Associate Professor Tachikawa et al. found that by preparing electrodes with mesocrystals doped (*5) with two different metal ions, it was possible to safely, cheaply and stably produce hydrogen peroxide as well as hydrogen. Hydrogen peroxide is used for many purposes including disinfecting, bleaching and soil improvement.
The research group’s next aim is to implement this technology. While continuing to improve the high efficiency of the developed photocatalyst electrode, they will try to assemble the cells into a compact module as a step towards societal implementation. They also plan to develop this mesocrystal technology with various materials and reaction systems.
This was a joint research project with Nagoya University’s Institute of Materials and Systems for Sustainability (Professor MUTO Shunsuke) and the Japan Synchrotron Radiation Research Institute (JASRI) (Chief Researcher OHARA Koji and Researcher INA Toshiaki).
The results were given advanced online publication in Nature Communications (Nature Publishing Group) on March 23, 2022.
Main points
- Hematite on its own is not suitable for producing hydrogen peroxide. By doping the hematite with different metal ions (tin and titanium) and sintering it, the researchers developed a highly active composite oxide co-catalyst (*6).
- The ability to produce hydrogen peroxide on-site in addition to hydrogen will contribute towards reducing the cost of solar water-splitting, as well as increasing the technology’s range of applications. Hydrogen peroxide is used for many purposes including disinfecting, bleaching and soil improvement.
Research Background
With the world facing increasing environmental and energy issues, hydrogen has gained attention as one of the possible next generation energy sources. Ideally, photocatalysts could use sunlight and water to produce hydrogen, however it is necessary to achieve a conversion rate of 10% to enable such a system to be adopted industrially. It has been pointed out that even if this efficiency is achieved, the cost of hydrogen will not reach the desired value. To overcome these issues, there is strong demand for the development of a competitive next generation solar water-splitting system with high added value that can produce other useful chemicals at the same time as hydrogen.
In their previous research, Tachikawa et al. developed ‘mesocrystal technology’, which involves precisely aligning nanoparticles in photocatalysts to control the flow of electrons and their holes. Recently, they have succeeded in dramatically increasing the light energy conversion efficiency by applying this technology to hematite.
Up until now, hematite has not been applied to the production of hydrogen peroxide. In this study, the researchers discovered that by modifying the surface of the hematite with a composite oxide of tin and titanium ions it was possible to produce both hydrogen and hydrogen peroxide in a highly efficient and selective manner.
Research Methodology
Mesocrystal technology:
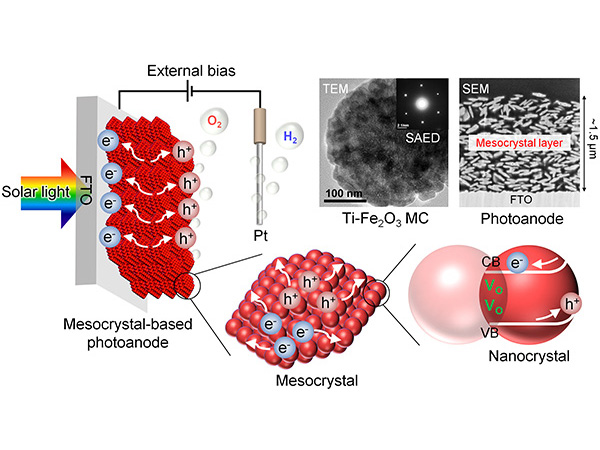
The main problem that causes a conversion rate decline in photocatalytic reactions is that the electrons and holes produced by light recombine before they can react with the molecules (in this case, water). Tachikawa et al. created 3D structures of hematite mesocrystals with highly oriented nanoparticles via solvothermal synthesis (*8). Furthermore, they were able to develop mesocrystal photoelectrodes for water splitting by coating and sintering the mesocrystals on the conductive glass substrate (Figure 1).
Formation of a co-catalyst for producing hydrogen oxide via dopant segregation:
Normally, photocatalytic water-splitting using hematite results in oxygen being produced from the oxidation of the water. Doping this hematite with tin ions (Sn2+) and titanium ions (Ti4+) and then sintering it at 700°C causes segregation of the tin and titanium dopants, leading to the formation of a composite oxide (SnTiOx) co-catalyst with high selectivity for hydrogen peroxide production (Figure 1). This structural change was revealed by performing synchrotron-based X-ray total scattering measurements using beamlines BL01B1 and BLO4B2 at the SPring-8 (*9) facility, and by using a high-resolution electron microscope incorporating electron energy loss spectroscopy (*10).
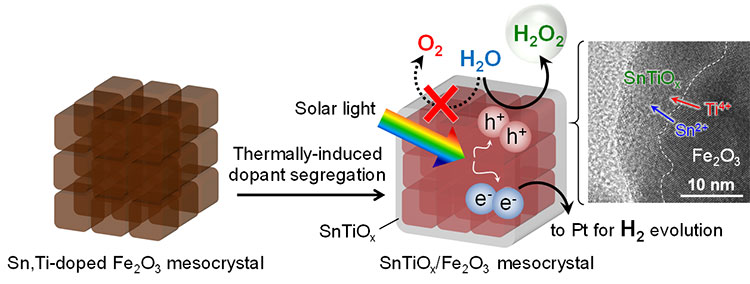
A hematite mesocrystal is a superstructure of particles, each around 20 nanometers in size. The mesocrystals were doped with Sn2+ and Ti4+ which were thermally induced to diffuse, segregating to form a composite oxide (SnTiOx) layer. The tin (Sn) on the uppermost layer is oxidized and becomes tin oxide (SnO2).
Photocatalyst formation and performance:
The water-splitting reaction was promoted when voltage was applied to the photocatalyst electrode illuminated by artificial sunlight (Figure 2a). The researchers investigated the photoelectric current density and the Faradiac efficiency (*11) which indicate the hydrogen production efficiency and the hydrogen peroxide selectivity, respectively. It was revealed that there were positive and negative effects on hydrogen and hydrogen peroxide production if the photocatalyst was doped with only one of the metal ions. On the other hand, hematite doped with both Sn2+ and Ti4+ could produce hydrogen and hydrogen peroxide at the same time in a highly efficient and highly selective manner. In addition, first principle calculations (*12) suggested that the SnTiOx co-catalyst on the hematite consisted of SnO2/SnTiO3 layers of a few nanometers in thickness (Figure 2b).
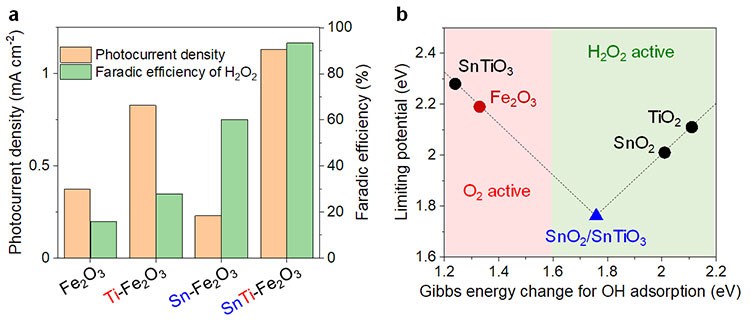
(a). The water-splitting properties of the dopant ions are different. The anode (*13) is a photocatalyst electrode, while the cathode (*14) is a platinum electrode. The potential is based on the RHE (Reversible Hydrogen Electrode) with 1.23 V being applied to produce oxygen gas from the water oxidation reaction. The photocatalyst electrode, which is doped with Ti4+, has a high hydrogen production rate but its hydrogen peroxide selectivity is low. If it is doped with Sn2+, its hydrogen peroxide selectivity increases while the amount of hydrogen it can produce decreases. By doping the catalyst with both Sn2+ and Ti4+ it is possible to achieve high selectivity and production rates for both hydrogen and hydrogen peroxide.
(b). Searching for high-activity co-catalysts using first principle calculations. Within the structure of the SnTiOx co-catalyst applied to facilitate hydrogen peroxide production, it was estimated that a SnO2 layer around 1 nanometer thick formed above the SnTiO3. The Gibbs energy change for OH absorption was close to 1.76 eV, indicating high photocatalytic activity for hydrogen peroxide production.
Further Developments
By modifying the surface of the hematite used for the photocatalyst, the research group succeeded in producing hydrogen peroxide, which hasn’t been produced in this manner before, in a highly efficient and selective way. Next, the researchers plan to further optimize the photocatalytic electrode and collaborate with industry to develop an onsite system for the production of hydrogen and hydrogen peroxide utilizing sunlight. They also plan to develop its applications to other metal oxides and reaction systems
Glossary
- 1. Hematite (α-Fe2O3)
- A type of iron oxide ore. In addition to being safe, inexpensive and stable (pH > 3), Hematite can absorb a wide range of visible light (approx. under 600nm).
- 2. Photocatalyst
- A material that can be utilized as a catalyst for reactions involving light illumination. The photocatalyst is applied to a conductive glass substrate (FTO glass) which absorbs the light. Used as an electrode, it can also be called a photocatalyst anode or a photoanode. In this study, a photocatalyst was used for the reaction to produce hydrogen by splitting the water molecules.
- 3. Hydrogen Peroxide
- Hydrogen Peroxide (H2O2) is commonly used for a wide range of applications, such as disinfectants, detergents, cosmetics, bleach and in purifying water. The majority of hydrogen peroxide is produced using the antraquinone process which must be conducted in a large-scale chemical plant and generates organic waste and CO2. In addition, hydrogen peroxide is unstable, therefore it is expensive to transport it and there are concerns about its safety. However, this research group developed a method of synthesizing liquid H2O2 via a safe, low-cost and green process. H2O2 has a higher market value than O2 so producing hydrogen peroxide at the same time as hydrogen can also reduce hydrogen production costs.
- 4. Mesocrystal
- Porous crystal structures consisting of nanoparticles that are three dimensionally aligned. Hundreds of nanometers or micrometers small, they feature pores between the nanoparticles that are between 2 to 50 nanometers.
- 5. Doping
- Adding a small quantity of another substance to the crystals to change their physical properties. Dopant diffusion occurs inside the crystal structure and the phenomenon whereby it is deposited on the surface is called dopant segregation.
- 6. Co-catalyst
- A substance which is combined with the photocatalyst to facilitate the reaction. In this study, a tin and titanium composite oxide was used to promote hydrogen peroxide production.
- 7. Light energy conversion efficiency
- The amount of light particles used in the reaction (output) divided by the amount of inputted light particles.
- 8. Solvothermal method
- A method of synthesizing solids using solvents at high temperatures and high pressures.
- 9. SPring-8
- Located in Harima Science Park in Hyogo Prefecture, Japan, SPring-8 is a large synchrotron radiation facility which currently provides the most powerful synchrotron radiation in the world. Synchrotron radiation is produced when electron beams, accelerated to almost the speed of light, are forced to travel in a curved path by a magnetic field, producing highly-focused powerful electromagnetic radiation. A wide range of research using synchrotron radiation is conducted at Spring-8, including nanotechnology, biotechnology and industrial applications. SPring-8 is managed by RIKEN, with the Japan Synchrotron Radiation Research Institute (JASRI) in charge of promoting its use.
- 10. Electron energy loss spectroscopy
- A spectroscopy technique to analyze the composition of a sample and bonding state of its elements by measuring the energy lost when the incident electron beam excites the electrons in the sample. By combining this technique with scanning transmission electron microscopy, it is possible to analyze minute regions at high resolutions.
- 11. Faradaic efficiency
- The percentage of the total electric current that is transferred into a system facilitating an electrochemical reaction (in this case the production of hydrogen and hydrogen peroxide).
- 12. First principle calculation
- A method of calculating the movement of electrons inside a substance, based on Density Functional Theory. It enables the properties for surface energy absorption and the optimum structure of a solid or the particles to be calculated.
- 13. Anode
- In electro-chemistry, the electrode where the oxidation reaction occurs
- 14. Cathode
- In electro-chemistry, the electrode where the reduction reaction occurs
Acknowledgements
This research was made possible thanks to the following organizations and grants:
- JST A-STEP Grant Number JPMJTR20TD.
- JSPS KAKENHI Grant Numbers JP18H01944, JP20H04673 and JP21H0204.
- MEXT’s program to promote research using the supercomputer Fugaku (Realization of innovative light energy conversion materials, Grant Number JPMXP1020210317).
- KANEKA CORPORATION (joint research and financial support)
- Thanks to JASRI, synchrotron radiation measurements could be conducted using SPring-8 beamlines BL01B1 and BL04B2 (under the proposals 2020A1208, 2021A1114, 2020A1209, and 2021A1113).
- Electron Microscope measurements were conducted with support from the Nagoya University microstructural characterization platform (one of MEXT’s Nanotechnology platform programs).
Journal Information
- Title
- “Binary dopant segregation enables hematite-based heterostructures for highly efficient solar H2O2 synthesis”
- DOI
- 10.1038/s41467-022-28944-y
- Authors
- Zhujun Zhang, Takashi Tsuchimochi, Toshiaki Ina, Yoshitaka Kumabe, Shunsuke Muto, Koji Ohara, Hiroki Yamada, Seiichiro L. Ten-no, Takashi Tachikawa
- Journal
- Nature Communications