Through single-cell RNA sequencing, a research group has successfully illuminated the characteristics of Myeloid immune cells in coronary plaque, which causes acute coronary syndrome (this term includes conditions such as unstable angina and acute myocardial infarction (heart attack)). Based on this data, they hope to develop a treatment method that can stabilize coronary plaque.
This study was conducted by a research group from Kobe University Graduate School of Medicine’s Division of Cardiovascular Medicine in the Department of Internal Medicine (Clinical Researcher Takuo Emoto, Associate Professor Tomoya Yamashita and Professor Kenichi Hirata) in collaboration with Hyogo Brain and Heart Center (one of Kobe University’s collaborative partners).
These research findings were published in Circulation on May 2, 2022.
Main points
- The characteristics of coronary plaques that cause acute heart attacks were revealed through single-cell RNA sequencing.
- More monocytes, mast cells and inflammatory macrophages (the latter of which exhibited higher expression of CXCL3 and IL1B) accumulated in coronary plaques involved in acute coronary syndrome in comparison to culprit plaques for chronic coronary syndrome (i.e. stable heart conditions).
Research Background
Recent decades have seen rapid advancement in analytical techniques. One such technique that has received much attention is single-cell RNA sequencing (*2), which enables the gene expression of each individual cell to be comprehensively analyzed. Single-cell RNA sequencing has been carried out on the plaque that causes carotid arteries to harden (arteriosclerosis), however no such studies have been conducted on carotid plaque due to it being very difficult to obtain a sample.
It was previously revealed that the formation of artery-hardening plaque is strongly linked to inflammation. However, the following points have not been fully understood: 1. What kind of immune cells are found in the carotid plaque of an actual human being, and what characteristics do these cells have? 2. Does carotid plaque act at the onset of acute coronary syndrome?
Research Methodology
Samples for this study were obtained by conducting directional coronary atherectomy procedures on consenting patients at Hyogo Brain and Heart Center between October 2022 and June 2021. Single-cell RNA sequencing was carried out on samples from 4 cases of chronic coronary syndrome and 3 cases of acute coronary syndrome. The results revealed the presence of the following myeloid cells in the plaque: macrophages (three types), monocytes, mast cells and dendritic cell clusters. In addition, a comparison of the results for the chronic cases with those of acute cases revealed that more monocytes, mast cells and inflammatory macrophages (the latter of which exhibited higher expression of CXCL3 and IL1B) accumulated in the coronary plaques in acute coronary syndrome cases (See Figure).
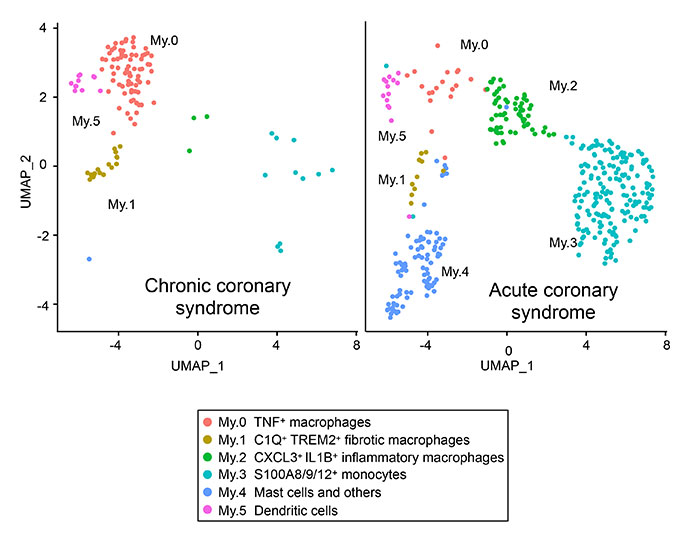
Comparison of single-cell RNA sequencing results for chronic coronary syndrome and acute coronary syndrome.
Further Developments
In a world first, this study revealed the distinct characteristics of myeloid immune cells in the coronary plaque of patients with acute coronary syndrome. Next, the researchers aim to use this data to develop a treatment method that can stabilize coronary plaque.
Glossary
- 1. Acute coronary syndrome
- This umbrella term describes a range of conditions that occur as a result of coronary plaque ruptures. These ruptures lead to blood clots that severely restrict or close the arteries. Examples of conditions included under the acute coronary syndrome term are unstable angina, acute myocardial infarction (heart attack) and sudden cardiac death.
- 2. Single-cell RNA sequencing
- A technique that allows the genetic expression of each individual cell to be comprehensively analyzed.
Journal Information
- Title
- “Single Cell RNA Seq Reveals a Distinct Immune Landscape of Myeloid Cells in Coronary Culprit Plaques Causing Acute Coronary Syndrome”
- DOI
- 10.1161/CIRCULATIONAHA.121.058414
- Authors
- Takuo Emoto , Hiroyuki Yamamoto, Tomoya Yamashita, Tomofumi Takaya, Takahiro Sawada, Shintaro Takeda, Masayuki Taniguchi, Naoto Sasaki, Naofumi Yoshida, Yoshihiro Saito, Tharini Sivasubramaniyam, Hiromasa Otake, Tomoyuki Furuyashiki, Clinton Robbins, Hiroya Kawai, Kenichi Hirata
- Journal
- Circulation
Regarding collaboration with a Kobe University-launched venture
Some of the pipelines for the single-cell RNA sequencing analysis in this study were set up in collaboration with Japan Research Activity Support inc. (JRAS).
JRAS is a Kobe University-launched venture that is supported by Kobe University Innovation. The research support gained through this industry-academia collaboration contributed towards making it possible to conduct the single-cell RNA sequencing in this study.