Autoimmune diseases cannot currently be cured, only treated, and this is also true for neuromyelitis optica spectrum disorder, which affects the central nervous system. A Kobe University study of how the treatment acts on the immune system shows that it shifts the balance of types of immune cells. This finding may represent a step toward the development of personalized medicine for autoimmune diseases.
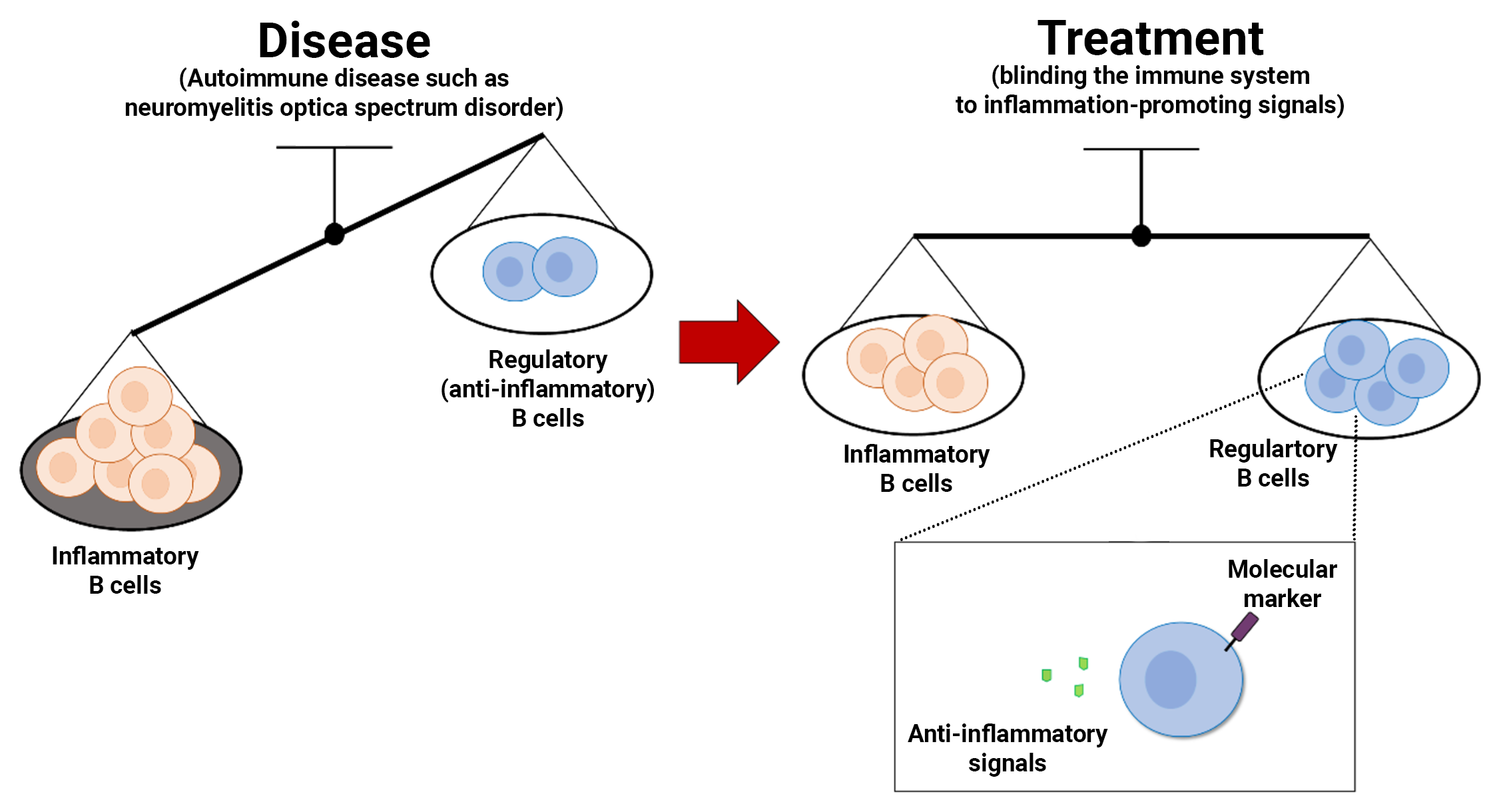
An autoimmune disease is the body’s immune system turning against parts of the body itself. Neuromyelitis optica disorder spectrum, or NMOSD, is one of them and it causes inflammation of the central nervous system, leading to vision and sensory loss, weakness and bladder dysfunction. The condition, which sometimes flares up in waves, has a treatment consisting of blinding the immune system to inflammation-promoting signals. But its biological action is broad and so it is also not understood why it doesn’t work in some patients and how to effectively ascertain which is the case.
The Kobe University neurologist CHIHARA Norio specializes on the disease and recently wondered: “B cells are a type of key immune cells that respond to inflammatory signals, and in autoimmune diseases like NMOSD, they produce antibodies against part of the body itself, exacerbating the condition. Therapies that inhibit inflammatory signals were therefore expected to change the activity of B cells in NMOSD. Since we observed that B cells were still present in the blood of patients after treatment, we decided to investigate the possibility that they were changing into a different type of B cell.” Chihara refers to so-called “regulatory B-cells,” a type of B cell that attenuates the immune system’s activity by secreting anti-inflammatory signal molecules and is thought to play an important role in keeping the body’s immune system from becoming too active, and in the case of autoimmune diseases may prevent disease flares.
The Kobe University team now reports that they created an experimental model of the immune cells during an NMOSD flare and could thus trace the effect of the drug on the different kinds of B cells. With the drug, there was a marked increase in the secretion of the anti-inflammatory signal, confirming their idea that not the number but the function of B cells responds to the treatment, according to the research published online on June 18, 2024, in Neurology® Neuroimmunology & Neuroinflammation, an official journal of the American Academy of Neurology.
In addition, Chihara’s team could also identify a molecular marker for B cells producing anti-inflammatory signals, a sort of ID that enables tracing the cells’ abundance. Having confirmed that healthy individuals and those under effective treatment show higher proportions of these cells than individuals in acute phases of the disease, Chihara thinks that this knowledge will enable clinicians to easily determine the effectiveness of the treatment in future diagnoses and thus is a step towards more personalized medicine.
Thinking about the bigger picture, the Kobe University neurologist thinks that this study will contribute to becoming able to not only treat but actually cure autoimmune diseases. He explains: “The essence of autoimmune diseases is a breakdown of autoimmune tolerance, the system that prevents us from attacking our own bodies. Our ultimate goal is to cure the disease by restoring this autoimmune tolerance, and the results of this study show one aspect of our work towards this goal.”
Acknowledgements
This research was funded by the Japan Society for the Promotion of Science (grants 21K20871, 23K14778, 20H03562 and 23H02797), the Japan Agency for Medical Research and Development (grant 22ek0109436h0003), the Japan Science and Technology Agency (grants JPMJMS2299 and JPMJMS229B), and the Ministry of Education, Culture, Sports, Science and Technology Japan (grants 22gm1710005h0001 and 23gm1710005h0002). It was conducted in collaboration with researchers from The University of Tokyo.
Original publication
R. Akatani et al.: Interleukin-6 signaling blockade induces regulatory plasmablasts in neuromyelitis optica spectrum disorder. Neurology® Neuroimmunology & Neuroinflammation (2024). DOI: https://doi.org/10.1212/NXI.0000000000200266