Polychlorinated biphenyls (PCBs) (*1) were widely used in industrial and commercial products including plastics, paints, electronic equipment and insulating fluids. Their manufacture was extensively banned from the late 70s onwards due to their toxicity, however large amounts still remain in our environment and accumulate inside animals’ bodies.
Chiral PCBs (*2) are PCBs that have two mirror-image isomers (*3); these isomers are identical reflections of each other with the same composition. Chiral PCBs are particularly dangerous because they have more chlorine atoms, which are hard for the body to break down, so they can accumulate in the body easily and their isomers are metabolized differently, causing isomer-specific toxicity (particularly neurodevelopmental issues). However, the process behind this selective metabolism was not known. To address this, a research group has illuminated how enzymes produced by the body unevenly metabolize the mirror-image isomers. These results will make it possible to estimate PCB metabolism and detoxification pathways in animals. They will also contribute towards the development of technology to make predictions about chiral PCBs’ mirror isomers, so that we can obtain a better understanding of potential toxicity in humans and other mammals.
These findings were made by a multi-institutional research collaboration, which included Associate Professor INUI Hideyuki (Kobe University Biosignal Research Center), Lead Researcher MATSUMURA Chisato (Hyogo Prefectural Institute of Environmental Sciences), Professor YAMAMOTO Keiko and Professor ITOH Toshimasa (Showa Pharmaceutical University), Associate Professor MORI Tadashi (Osaka University Graduate School of Engineering), and Visiting Professor NAKANO Takeshi (Osaka University Research Center for Environmental Preservation).
These research results were published online in the international academic journals Environmental Science & Technology on July 8, and Chemosphere on September 6, 2022.
Main points
- In the past, PCBs were utilized in a vast range of industrial and commercial products. These highly carcinogenic chemical compounds remain in our environment and accumulate inside organisms.
- PCBs have a dioxin-like toxicity and research into PCB metabolism is advancing.
- However, research had yet to uncover how chiral PCBs’ mirror-image isomers are metabolized.
- The researchers split the two atropisomers (mirror-image isomers) found in each type of chiral PCB and used them as substrates for CYP enzymes (*4).
- Even though a pair of atropisomers are physically and chemically identical, there was a big difference in the extent to which they were metabolized.
- Differences in CYP’s amino acids’ binding inhibition of each atropisomer cause the atropisomers to be metabolized differently.
- These findings will be useful for measuring the atropisomers of chiral PCBs, which accumulate easily inside animals’ bodies.
Research Background
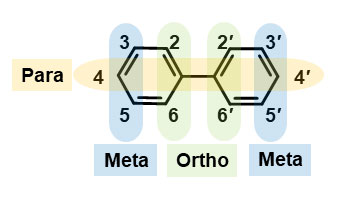
Even though the manufacture and use of PCBs was banned around 50 years ago, they still remain in the environment. It has been discovered that PCBs accumulate inside the bodies of humans and other animals through food consumption. In particular, PCBs with many chlorine bonds are water resistant and do not break down easily. This enables high concentrations of these PCBs to accumulate inside animals’ bodies, which adversely affects their health. PCBs’ toxicity is induced by the aryl hydrocarbon receptor (AhR) (*5), causing similar adverse effects to dioxin (*6) poisoning such as cancer, teratogenesis and immune system damage. Research is being conducted on the particular types of PCB widely known to cause these effects, which are dioxin-like PCBs with one ortho chlorine substitution in the biphenyl ring of their chemical structure, or PCBs with no substitutions (Figure 1). However, if a PCB has more than 3 chlorine substitutions at the ortho position of the biphenyl ring, it becomes a mirror-image isomer called chiral PCB (Figure 2). These chiral PCBs do not demonstrate dioxin-like toxicity but are far more dangerous, binding with the ryanodine receptors (RyR) in organisms to become neurotoxic. The two mirror-image isomers (called atropisomers) in chiral PCB have identical physical and chemical properties and exist at a 1:1 ratio in commercial chiral PCB. However, biased ratios are often observed in the environment and in animals such as earthworms and whales, as well as humans. It is believed that this unbalanced ratio is mainly caused by metabolism and that one of chiral PCB’s atropisomers is more effected by the metabolic reaction thus reducing its concentration.
However, very little research has been carried out into differences in how these atropisomers are metabolized nor the structural arrangement of the metabolic enzymes.
Research Methodology
To address this knowledge gap, the team conducted research focusing on the metabolic enzyme cytochrome P450 (CYP enzyme). The CYP enzyme reacts with foreign compounds that enter an animal’s body (for example, chemicals or pollutants in food, or medicines). CYP can convert them into water-soluble compounds and promote their expulsion from the body. Previous research by this group has shown that CYP enzymes hydroxylate and dechlorinate dioxin-like PCBs. This decreases PCB’s binding with AhR and increases its water solubility, promoting expulsion from the body and therefore counteracting its toxicity. In other words, CYP is an important enzyme that determines whether or not PCBs are treated as toxic compounds by the body. To measure the metabolic action of CYP on chiral PCB, the researchers set up a CYP enzyme and PCB docking model. They used this to estimate the structure of PCB metabolites and the structure of the CYP that decides to metabolize each of the PCB atropisomers differently.
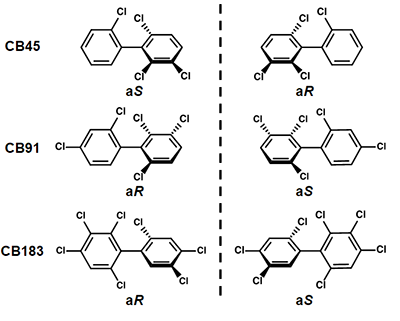
For the experiment, the group selected three types of chiral PCB, each with a different number of substituted chlorine atoms; CB45 (4 chlorine substitutions), CB91 (5 chlorine substitutions) and CB183 (7 chlorine substitutions) (Figure 2). They separated the atropisomers for each type of chiral PCB using chromatography and let them react with a human CYP enzyme. It is thought that research on separating the atropisomers and letting them react has not been done before now. The results revealed big differences in how each atropisomer is metabolized (Figure 3). This revealed that even though the two atropisomers in one PCB have the same physical and chemical composition, they are biologically different. The researchers found that one of the chiral PCB atropisomers was metabolized more than the other one, disrupting the 1:1 ratio. In addition, it is thought that the amount of (aS)-CB183 atropisomer decreases because it is metabolized more than the other atropisomer, and this is supported by the reports of low accumulation of (aS)-CB183 in humans.
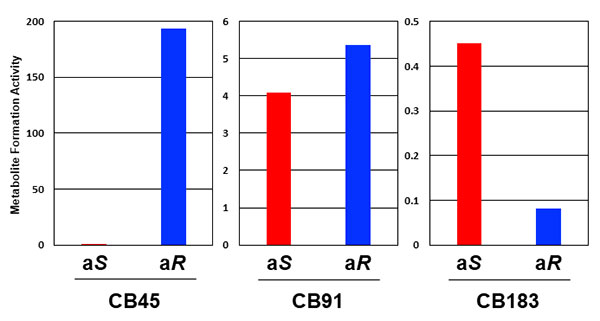
Of the two atropisomers in each chiral PCB, (aR)-CB45 in CB45 and (aS)-CB183 in CB183 are metabolized into hydroxylated metabolites more. In CB91, each atropisomer is metabolize dat a similar rate.
But why are these physically and chemically identical atropisomers metabolized differently by the CYP enzyme? To solve this mystery, the researchers used a computer model to investigate how easily each chiral PCB atropisomer binds to the chemical structure of CYP. They found that when an atropisomer fills up the substrate-binding cavity inside the CYP enzyme, CYP’s amino acids (that form the cavity) interfere with the binding between CYP and the atropisomer (see Figures 4 and 5).
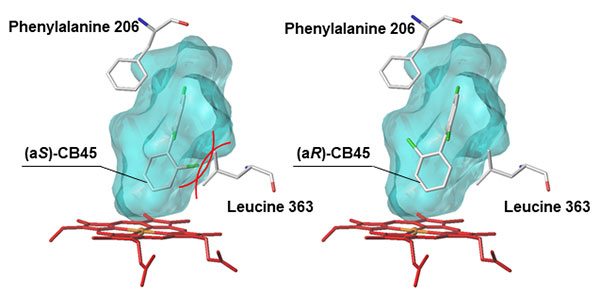
Light blue indicates the substrate-binding cavity inside the CYP enzyme.The CYP enzyme is unable to form a stable interaction with the atropisomer (aS)-CB45 because (aS)-CB45 collides (indicated by the red lines) with leucine 363, therefore CYP produces fewer metabolites (see Figure3).
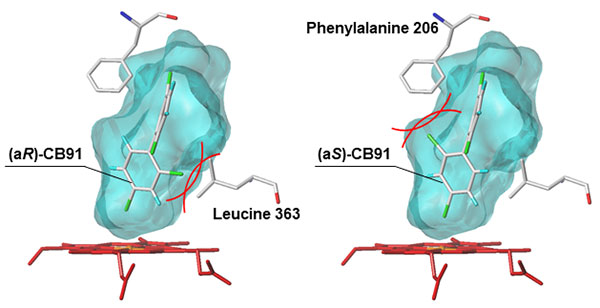
Therefore, the atropisomer that isn’t interfered with by CYP’s amino acids becomes easy to metabolize (atropisomer (aR)-CB45 in CB45, and (aS)-CB183 in CB183), resulting in alterations to the original 1:1 ratio of atropisomers found in chiral PCB.
Further Research
The results of this research will be useful for making predictions about the atropisomers of chiral PCBs, which accumulate easily inside animals’ bodies. In other words, it will be possible to work out which atropisomer is reduced by the metabolic reaction with CYP enzymes and which atropisomer remains inside the body. Chiral PCB’s toxicity is activated by binding with RyR, however the ability to bind with RyR differs between the atropisomers. Therefore, this research will make it possible to estimate the toxicity of chiral PCBs.
Glossary
1. Polychlorinated biphenyl (PCB)
A chemical compound with between 1 to 8 chlorine atoms bound to its biphenyl ring. There are 209 types of PCB, each with different structures. Until the late 1970s, PCBs were manufactured for use as insulating oil and were also found in a variety of products, however their manufacture and use were banned worldwide after it was discovered that they are highly toxic. PCBs with many chlorine bonds do not break down easily so large quantities remain in the environment and inside organisms even almost 50 years after they were banned.
2. Chiral PCB
This is a PCB that has 3 or more chlorine substitutions at the ortho position. This structure inhibits the rotation of the bond connecting the 2 benzene rings. It also has 2 mirror isomers (a pair of atropisomers). There are 19 types of chiral PCB and according to the Cahn-Ingold-Prelog priority rules, their atropisomers are referred to as ‘aS’ and ‘aR’.
3. Mirror-image isomer (enantiomer)
A pair of isomers that are non-superimposable mirror-images of each other, slightly similar to your left and right hands. Asymmetrical carbon compounds have mirror-image isomers. These isomers have the same physical and chemical properties. In chemical synthesis, a racemic mixture consists of an equal amount of each isomer in a 1:1 ratio.
4. Cytochrome P450 monooxygenase (CYP enzyme)
This metabolic enzyme is found in most organisms- it is responsible for the oxygenation reaction in the biosynthesis pathway for various chemical compounds in the body. In addition to this it has a detoxifying function; it oxygenates foreign compounds in the body, which makes them water soluble and easier to excrete.
5. Receptor
These are proteins found in organism cells that bind to chemical substances and promote or control the expression of the gene required for the organism to respond to the chemical substance. Aside from the estrogen receptor (ER) that binds to the female hormone and the aryl hydrocarbon receptor (AhR) that binds to dioxins, there are many other known receptors for various hormones.
6. Dioxin
Dioxins are generated by incinerating trash at low temperatures. They are difficult to break down and persist in the environment and inside organisms for a long period of time. When dioxins enter an animal’s body via food, they bind to AhR (aryl hydrocarbon receptors) inside the cells and the gene transcription for the enzyme that detoxifies dioxins is activated.
Acknowledgements
This study was supported by a Grant-in-Aid for Challenging Exploratory Research (grant number 25550064) from the Japan Society for the Promotion of Science and the Japan Science and Technology Agency’s CREST program (grant number JPMJCR2001).
Journal Information
Title
DOI
10.1021/acs.est.2c01155
Authors
Hideyuki Inui1,2, Terushi Ito2, Chiharu Miwa3, Yuki Haga4, Makoto Kubo5, Toshimasa Itoh5, Keiko Yamamoto5, Masayuki Miyaoka6, Tadashi Mori6, Harunobu Tsuzuki2, Shintaro Mise2, Erika Goto2, Chisato Matsumura4, and Takeshi Nakano7
- Biosignal Research Center, Kobe University.
- Graduate School of Agricultural Science, Kobe University.
- Faculty of Agriculture, Kobe University.
- Hyogo Prefectural Institute of Environmental Sciences, Hyogo Environmental Advancement Association.
- Showa Pharmaceutical University.
- Graduate School of Engineering, Osaka University.
- Research Center for Environmental Preservation, Osaka University.
Journal
Environmental Science & Technology
Title
DOI
10.1016/j.chemosphere.2022.136349
Authors
Terushi Ito1, Chiharu Miwa2, Yuki Haga3, Makoto Kubo4, Toshimasa Itoh4, Keiko Yamamoto4, Shintaro Mise1, Erika Goto1, Harunobu Tsuzuki1, Chisato Matsumura3, Takeshi Nakano5, Hideyuki Inui1,6
- Graduate School of Agricultural Science, Kobe University.
- Faculty of Agriculture, Kobe University.
- Hyogo Prefectural Institute of Environmental Sciences, Hyogo Environmental Advancement Association.
- Showa Pharmaceutical University.
- Research Center for Environmental Preservation, Osaka University.
- Biosignal Research Center, Kobe University.
Journal
Chemosphere
Related link