Applying ice to a muscle injury is a widespread first-aid treatment, but exactly what effect does this have on the muscle regeneration and does it really help? Cumulative research by a multi-institutional Japanese research collaboration reveals that ‘to ice or not to ice’ may depend on the degree of muscle injury.
In their latest research, the group consisting of Associate Professor ARAKAWA Takamitsu and Master’s student NAGATA Itsuki (from Kobe University’s Graduate School of Health Sciences), and Assistant Professor KAWASHIMA Masato (Kawasaki University of Medical Welfare) et al. have shown that applying ice to muscle damage in a small percentage of muscle fibers in rats promotes muscle regeneration. This is believed to be the first study in the world to show benefits of icing on muscle repair. In conjunction with their previous study on serious muscle injuries (‘Icing muscle injuries may delay recovery’), it is hoped that these results can be used as a basis for more accurate guidelines on whether or not to ice such injuries.
These research findings were first reported in the American Journal of Physiology-Regulatory, Integrative and Comparative Physiology on March 6, 2023.
Main points
- The experiments showed that applying ice after a mild muscle injury promotes muscle regeneration.
- This is believed to be the first time in the world that a study has shown a positive effect of icing on muscle regeneration.
- The researchers showed that the extent of the injury may have a greater impact on the effectiveness of icing than the method or timing employed.
- The findings of this ongoing research will lead to the spread of more accurate information on the effects of icing throughout hospitals, and in the realms of sports and physical education.
Research Background
‘RICE treatment’ is a common approach for treating the acute phase of sports injuries. This acronym stands for Rest, Ice, Compression and Elevation and it is also often used in physical education in schools and even clinical settings. There are a variety of subsequent steps that can be taken to treat the injury afterwards, yet opinions vary as to whether or not icing should be applied. However, there is a lack of evidence on the benefits of icing.
The current research team has conducted many experiments to investigate the effectiveness of icing, which led them to publish their previous findings (‘Icing muscle injuries may delay recovery’). However, no previous animal experiments have indicated that icing promotes muscle regeneration.
In this study, the researchers focused on altering the severity of the muscle injury in the experiments. The reasoning behind this was that the majority of sports-related muscle injuries are limited; in other words less than 10% of the overall number of muscle fibers (myofibers) are damaged and necrotized. However, all animal experiments up until now had looked at more serious injuries where over 20% of the myofibers were damaged.
Thus, the team devised an animal model for mild muscle injuries, and experimented with applying ice after injury using a similar method as before.
Research Findings
After the animal was anaesthetized, the muscle was exposed and clamped between forceps to induce injury. In their previous experiments, the researchers attached a 500g weight to the forceps, which induced an injury that affected 20% of the total number of fibers in the muscle. In the present study, they tried attaching a 250g weight to the forceps and demonstrated that this could be used to consistently injure 4% of the fibers (Figure 1). This is similar to the degree of injury that often occurs after sports activities such as vigorous exercise or long-distance marathon running.
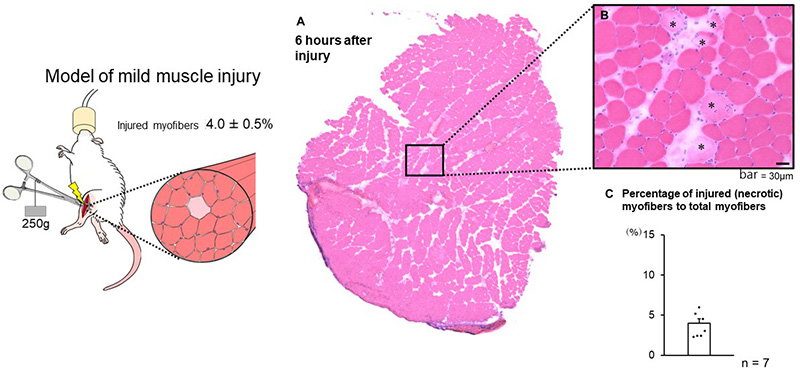
Left: Method of inducing mild muscle injury. While under anaesthetic, the protractor muscle in the animal’s hind leg is exposed and then clamped by forceps with a 250g weight attached.
Right: The muscle injuries induced using this method affect around 4% of total myofibers. C Shows that this method enables mild injury to be consistently induced.
Icing was carried out by placing polyethylene bags of ice on surface of the skin over three 30-minute sessions per day, with each session being 1.5 hours apart. This was continued until two days after injury for a total of 9 icing sessions (i.e. immediately after injury = 3 sessions, 1 day after injury = 3 sessions, 2 days after injury = 3 sessions). The icing method was the same as in the previously reported study (‘Icing muscle injuries may delay recovery’).
Observations of muscles that were regenerating in the icing group and no-icing group 2 weeks after injury revealed significant differences in the size of regenerating fibers in cross-sections (Figure 2). In other words, this demonstrated the possibility that skeletal muscle regeneration is promoted by icing.
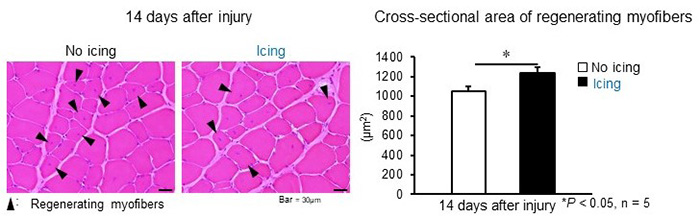
Left: Cross-section of muscles two weeks after injury. The muscle from the animal on the left received no treatment while the muscle on the right received icing treatment.
Right: Comparison of cross-sectional area of regenerating myofibers. 2 weeks after mild muscle injury, the cross-sectional area of regenerating myofibers was significantly larger for muscles that were iced (black bar) than for muscles that received no treatment (white bar).
Macrophages are immune cells that orchestrate the reparative process of injured muscle. Pro-inflammatory macrophages accumulate in the damaged site soon after injury occurs, however they express an inducible nitric oxide synthase (iNOS), which has a disadvantageous side-effect of expanding the injury’s size. The results of this team’s experiments revealed that icing after mild muscle injury reduces the accumulation of iNOS-expressing pro-inflammatory macrophages (Figure 3). By causing this phenomenon, icing prevents the expansion of muscle injury size (Figure 3).
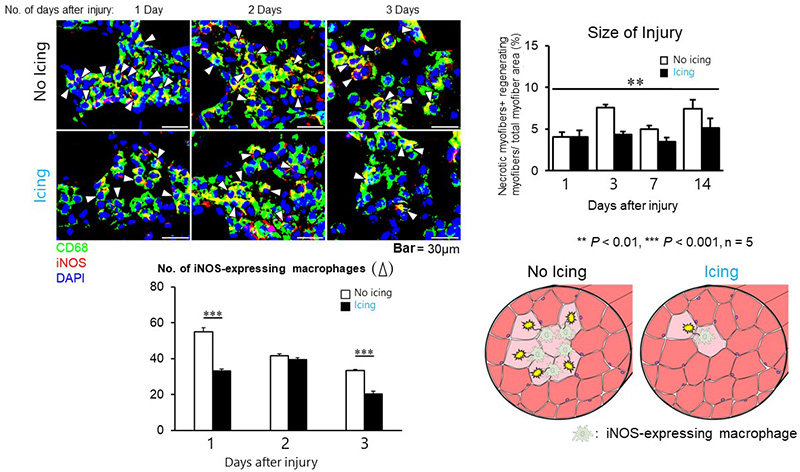
Top left: Cross-sections of muscles from animals given either no treatment or icing treatment after mild muscle injury. Comparison of inflammatory macrophage distribution (indicated by white arrowheads) from 1 day after injury through to 3 days after injury. On day 1 and day 3 in the icing group, hardly any pro-inflammatory macrophages accumulated in the injury site.
Bottom left: Number of pro-inflammatory macrophages in the field of view. There were few inflammatory macrophages at the site of injury if icing was carried out.
Top right: Injury size comparison. The percentage of total myofiber area taken up by injured fibers was compared. Expansion of injury was not found in muscle from the icing group (black bars).
Bottom right: Schematic diagrams of the phenomena occurring inside untreated injured muscles and injured muscles given an icing treatment.
In other words, icing attenuates the recruitment of pro-inflammatory macrophages in the injury site. This was also reported in their previous study (‘Icing muscle injuries may delay recovery’), demonstrating that this is an effect caused by icing regardless of whether the muscle injury is serious or mild. In the previous study, icing was found to delay the regeneration of muscle after a serious injury that destroyed many fibers because the pro-inflammatory macrophages were unable to sufficiently phagocytose (*5) the injured muscle. In contrast to this, the current study shows that icing has a positive effect when the muscle injury is mild because it prevents the secondary expansion of the muscle injury caused by the pro-inflammatory macrophages. It suggests that this particular effect of icing is connected to the promotion of muscle regeneration.
Further Research
Icing has been used in the treatment of muscle injuries for a long time, however the positive effects of icing had yet to be elucidated until now. This study has shown that icing can promote muscle regeneration when used to treat commonly-occurring mild muscle injuries.
However, this does not mean that icing is effective for all types or degree of muscle injury. The researchers aim to further elucidate and raise awareness of this. For example, the group’s previous study showed that icing actually inhibited regeneration in cases of serious muscle injury. In addition, the term ‘muscle injury’ also includes extremely minute injuries that have yet to be observed through the team’s animal experiments, so it is still unclear as to what effect icing has on the repair from such microtraumas.
The researchers’ next challenge is to determine the extent of muscle injury up to which icing is appropriate. By building upon their previous investigations, they aim to contribute towards guidelines that will enable people in sports and clinical rehabilitation to make accurate judgements about whether or not to ice an injury.
Glossary
- *1 Macrophage
- A population of white blood cells. There are two known types of macrophage: pro-inflammatory and anti-inflammatory.
- *2 Pro-inflammatory macrophage
- A type of macrophages that immediately migrate into the site of a tissue injury. They phagocytose the damaged tissue and trigger inflammation.
- *3 Inflammation
- A pathological reaction that occurs when tissue is damaged. Symptoms include rubefaction (reddening of the skin), feverishness, swelling and pain.
- *4 Inducible nitric oxide synthase (iNOS)
- An enzyme involved in the synthesis of nitric oxide. Macrophages can damage muscle cells when they express iNOS.
- *5 phagocytose (noun = phagocytosis)
- A behavior of macrophages that surrounds and removes damaged tissue.
Acknowledgements
This study received support from the following:
- Japan Society for the Promotion of Science (JSPS KAKENHI grant number: 21K11238)
- Yamaha Motor Foundation for Sports
- Wesco Scientific Promotion Foundation
Journal Information
- Title
- “Icing after skeletal muscle injury with necrosis in a small fraction of myofibers limits iNOS-expressing macrophage invasion and facilitates muscle regeneration”
- DOI
- 10.1152/ajpregu.00258.2022
- Authors
- Itsuki Nagata*, Masato Kawashima*, Anna Miyazaki, Makoto Miyoshi, Tohma Sakuraya, Takahiro Sonomura, Eri Oyanagi, Hiromi Yano, Takamitsu Arakawa
*co-first authors - Journal
- American Journal of Physiology-Regulatory, Integrative and Comparative Physiology