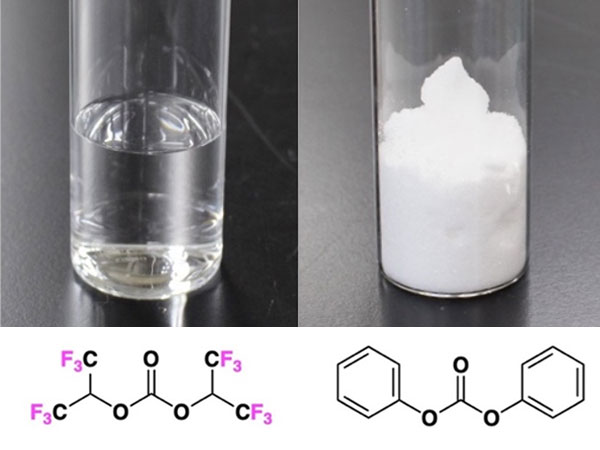
The widely used dry-cleaning and degreasing solvent perc can be converted to useful chemicals by a new clean, safe and inexpensive procedure. The Kobe University discovery using on-demand UV activation may open the path to upcycling perc and thus contribute to a more sustainable society.

Photo-on-demand one-pot synthesis provides an environment in which harmless source materials such as the dry-cleaning agent perc can be upcycled to useful products safely, cheaply and in an environmentally friendly way. © TSUDA Akihiko (CC BY)
Organic synthesis is the production of useful chemicals, such as drugs, from other available chemicals. In general, chemists use source materials to create simple building blocks, such as carbonate esters, and combine them to increasingly complex structures. The source materials for this need to be reactive, but that also usually makes them toxic, such as the commonly used phosgene. For this reason, chemists are constantly on the lookout for new source materials that are safer and more environmentally friendly while also retaining their reactivity. Ideally, they would like to use abundant and safe waste materials and turn them into useful products without that process also resulting in toxic by-products.
The Kobe University research group of TSUDA Akihiko specializes in the development of one-pot and flow organic synthesis using UV light. Such reactions have the advantages that they provide a closed environment in which harmless source materials can be photo-activated by the high-energy light on demand, eliminating the need for procuring or storing possibly toxic source materials; and that any reactive and thus possibly dangerous products can immediately be reacted further with other compounds, eliminating the need to extract them or the danger of their leaking into the environment. They now built on their experience and partnered with the Japanese material manufacturer AGC Inc. to refine their process. Together, they focused on an attractive target, the commonly used degreasing and dry-cleaning agent perc, or perchloroethylene as chemists call it, which is non-harmful and produced in large quantities around the world.
Publishing in The Journal of Organic Chemistry, the academia-industry partnership describes a method to turn perc into carbonate esters and chloroform, valuable building blocks for further organic synthesis, efficiently and in large quantities without any direct handling of toxic source materials such as phosgene. Tsuda explains: "Because perc is nonflammable and stable enough to be used as a solvent, its use as a raw material for organic synthesis has received little attention. However, using our original photo-on-demand organic synthesis method, we have succeeded for the first time in simultaneously obtaining industrially important carbonates and chloroforms from that source."
In addition, to further reduce the environmental impact of their system, they tested replacing conventional mercury lamps that emit high-energy UV light with LED lamps generating mild UV light. While that required some modification of the reaction process, they could still successfully synthesize the desired products, opening up even greater potential to make organic synthesis more sustainable.
Tsuda is hopeful for the environmental impact of their new development. He says, "It is a safe, inexpensive, simple, and environmentally friendly chemical reaction. I expect that this new method of utilizing and upcycling perc, which is used in large quantities around the world, will be a significant step toward the realization of a carbon neutral and sustainable society."
Acknowledgements
This research was supported by the Adaptable and Seamless Technology Transfer Program through Target-driven R&D (A-STEP) from the Japan Science and Technology Agency. It was conducted in collaboration with AGC Inc.
Original publication
I. Higashimura et al.: Photo-on-Demand In Situ One-Pot Synthesis of Carbonate Esters from Tetrachloroethylene. The Journal of Organic Chemistry (2024). DOI: 10.1021/acs.joc.3c02588